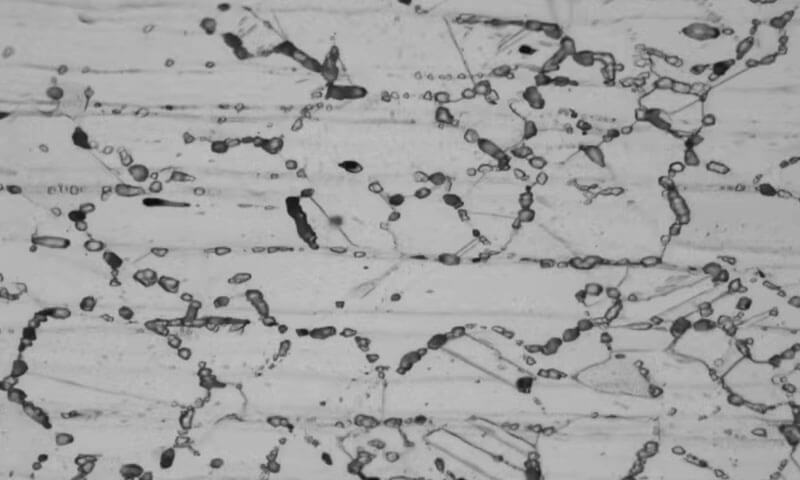
Panimula
Intergranular kaagnasan (IGC), also called intergranular attack (IGA), is a localized form of corrosion that progresses preferentially along grain boundaries rather than through the grain interiors.
Sa praktikal na mga termino, the metal may appear acceptable at the surface while a narrow network of attack develops beneath it, eventually reducing strength and causing separation, grain detachment, or failure.
Grain boundaries are inherently higher-energy regions, but they usually do not become a corrosion problem unless alloy chemistry or thermal history makes them chemically different from the surrounding matrix.
1. Definition of Intergranular Corrosion
A rigorous definition is straightforward: intergranular corrosion is corrosion that occurs at and adjacent to grain boundaries, with comparatively little attack in the grain interiors.
In the simplest electrochemical picture, the grain-boundary region becomes the anodic site and the grain interior acts as the cathode, so the corrosion path follows the boundary network.
That boundary attack becomes especially dangerous when the grain boundaries are chemically altered by precipitation or segregation.
For stainless steels, ASTM A262 identifies susceptibility to intergranular attack in austenitic grades by multiple standardized tests,
and it explicitly associates acceptable oxalic-acid etch behavior with freedom from susceptibility linked to chromium carbide precipitation.

2. Formation Mechanism of Intergranular Corrosion
The central mechanism is grain-boundary chemistry change.
During sensitization or aging, alloying elements or impurities can precipitate at the grain boundaries, or protective elements can be depleted from the adjacent matrix.
Once that happens, the boundary region and the surrounding grain no longer share the same electrochemical potential, and the boundary becomes the preferred site for dissolution.
In austenitic stainless steels, the classic mechanism is chromium carbide precipitation at grain boundaries.
The chromium consumed by carbide formation leaves a chromium-depleted zone next to the boundary, and that depleted band loses enough corrosion resistance to become preferentially attacked.
ASTM A262 treats this as the standard sensitization-related problem in austenitic stainless steels, and ASTM G108 uses electrochemical reactivation to quantify the degree of sensitization in Type 304 and 304L.
Para sa aluminyo alloys, the mechanism is different in detail but similar in structure: grain-boundary precipitates and adjacent precipitate-free zones create local microgalvanic cells.
The precipitates, the PFZ, and the matrix can end up with different compositions and corrosion potentials, which makes the grain boundary the preferred corrosion path.
Published work on age-hardenable aluminum alloys shows that quench rate is a major processing variable because it affects boundary segregation and the size/distribution of grain-boundary precipitates.
3. Causes of This Type of Damage
Intergranular corrosion usually does not arise from a single cause. It develops when several conditions combine:
- a susceptible alloy chemistry,
- a thermal cycle that allows grain-boundary precipitation or segregation,
- insufficient cooling rate or inappropriate heat treatment,
- and an environment that can exploit the weakened boundary region.
In stainless steels, low carbon content helps because it reduces the available carbon for chromium carbide formation, and stabilized or extra-low-carbon grades are designed to resist sensitization during ordinary welding operations.
ASTM A262 specifically notes that extra-low-carbon grades and stabilized grades such as 304L, 316L, 317L, 321, at 347 are tested after sensitizing heat treatments in the range where carbide precipitation is most likely.
In aluminum alloys, the important cause is the combination of solute segregation, precipitate formation, and PFZ development around grain boundaries during solution treatment, pagpapawi ng, and aging.
Water quenching after solution treatment can prevent intergranular corrosion susceptibility in some age-hardenable aluminum alloys by limiting harmful boundary precipitation and segregation.
In duplex stainless steels, long-term aging can promote phase changes such as sigma-phase growth, which increases sensitization and lowers breakdown potential.
Recent work on lean duplex stainless steel shows that aging at 700 °C at 800 °C changes the intergranular corrosion response through phase evolution and self-healing behavior.
4. Materials Susceptible to Intergranular Corrosion
| Material family | Typical susceptibility mechanism | Why it is vulnerable | Common control strategy |
| Austenitic hindi kinakalawang na asero | Chromium carbide precipitation and chromium depletion at grain boundaries. | Sensitization creates a chromium-depleted zone that loses passivity. | Mababang-carbon na grado, stabilized grades, solusyon annealing, mabilis na paglamig, weld control. |
| Ferritic hindi kinakalawang na asero | Chromium carbide or nitride precipitation during inappropriate thermal exposure or welding. | Boundary precipitation can create locally weaker corrosion resistance. | ASTM A763 screening, heat-treatment control, weld procedure control. |
| Duplex stainless steels | Phase imbalance and secondary-phase formation during aging or welding. | Sigma phase and other transformations can increase sensitization and lower resistance. | Tight thermal control, balanced ferrite/austenite, postweld treatment where needed. |
Age-hardenable aluminyo mga haluang metal |
Grain-boundary precipitates and PFZ microgalvanic coupling. | Boundary chemistry differs from matrix chemistry, enabling preferential attack. | Control solution treatment, quench rate, and aging condition. |
| Nickel-base alloys | Grain-boundary carbides and intermetallic phases, especially after poor thermal control. | Boundary precipitation can degrade corrosion resistance and weld-zone performance. | Alloy selection, heat-input control, and appropriate postweld practices. |
| tanso in certain conditions | Boundary enrichment or segregation, including zinc-related effects. | Boundary chemistry can become more reactive than the grains. | Alloy selection and environment control. |
5. Hazards of Intergranular Corrosion
Intergranular corrosion is dangerous not because it always looks severe, but because it often develops in a way that is structurally hidden.
The metal may retain its surface appearance for a long time while its grain boundaries are quietly being weakened.
Once the boundary network is sufficiently attacked, the component can lose ductility, lakas ng loob, pressure tightness, and fatigue resistance far earlier than expected.
This is what makes intergranular corrosion especially treacherous in critical equipment.
Loss of Mechanical Integrity
The most direct hazard of intergranular corrosion is the gradual loss of load-bearing capability.
Because the attack progresses along grain boundaries, the metal can suffer a significant reduction in effective cross-section and cohesion without showing the uniform thinning typical of general corrosion.
This is particularly serious for components that depend on:
- lakas ng paghatak,
- bending resistance,
- pressure containment,
- or cyclic load capability.
A part affected by intergranular corrosion may still look intact during inspection, yet its internal grain-boundary network may already be severely compromised.
When the material is later loaded, the weakened boundaries can separate with little warning.
Sudden and Brittle-Type Failure
Intergranular corrosion often converts a normally ductile material into one that fails in a much more brittle manner.
Once the grain boundaries lose cohesion, cracks can propagate rapidly along the weakened network.
The result is often a fracture surface that appears granular or intercrystalline rather than smoothly ductile.
This hazard matters because it reduces the margin for warning. Instead of slow, visible wall thinning, the component may fail after only modest additional loading or vibration.
Sa pagsasanay, this makes intergranular corrosion one of the more dangerous localized corrosion modes in terms of unexpected failure.
Leak Formation and Pressure-Boundary Failure
For pipes, mga tangke, mga heat exchanger, mga katawan ng balbula, and welded pressure equipment, the main concern is often not only strength loss but also loss of tightness.
Intergranular corrosion can create a boundary-connected network of microcracks and voids that eventually allows fluid leakage.
This is especially hazardous in systems carrying:
- corrosive liquids,
- pressurized gases,
- hot process streams,
- or hazardous chemicals.
A component may remain dimensionally sound enough to pass casual visual checks, but still fail as a pressure boundary because the corrosion has created a path for leakage along the grain boundaries.
Rapid Crack Propagation Under Stress
Once intergranular attack has progressed, any service stress can accelerate damage.
Vibrations, thermal cycling, mechanical shock, and residual stress all help open already-weakened grain boundaries.
This is why intergranular corrosion is often paired with secondary cracking problems such as stress-assisted fracture.
The danger is not just the corrosion itself, but the interaction between corrosion and load.
A component may survive in a benign stress state yet fail quickly when the same corrosion-damaged microstructure is exposed to real operating forces.
Reduced Fatigue Life
Components exposed to repeated loading are especially vulnerable because grain-boundary attack creates small crack initiators.
These sites concentrate stress and reduce the number of cycles the material can survive before failure.
The fatigue hazard is significant in:
- rotating shafts,
- cyclic pressure vessels,
- Mga Welded na Istraktura,
- mga bukal,
- and machine parts exposed to vibration.
Sa gayong mga kaso, intergranular corrosion does not merely shorten life; it can completely change the failure mode from predictable fatigue accumulation to premature fracture.
Loss of Ductility and Toughness
A material that has suffered grain-boundary attack may still have acceptable nominal chemistry, but its ductility and toughness can be sharply reduced.
That makes it less capable of absorbing impact, thermal distortion, or local overload.
This is especially problematic after fabrication, repair welding, or heat exposure, because the damaged region may be expected to behave like the rest of the component.
Sa katotohanan, the corrosion-altered grain boundaries can create a mechanically weak zone that behaves very differently from the unaffected base metal.
6. Control Measures
Preventing intergranular corrosion is not a single-action problem.
It requires control at four levels at once: pagpili ng haluang metal, thermal history, fabrication practice, and service environment.
If any one of these is neglected, the grain-boundary condition can become chemically unstable and the material may remain vulnerable even when the bulk alloy appears sound.
Pagpili ng Materyal: Prevent the Problem at the Design Stage
The first and most effective control measure is choosing an alloy that is inherently less susceptible to grain-boundary attack in the intended environment.
Use low-carbon grades where sensitization is a risk
For stainless steels, low-carbon grades such as 304L, 316L, and similar extra-low-carbon variants are preferred when welding or elevated-temperature exposure is expected.
Lower carbon reduces the amount of carbide that can form at grain boundaries, which in turn reduces chromium depletion and the associated corrosion risk.
Use stabilized grades for demanding thermal service
Grades stabilized with titanium or niobium, tulad ng 321 at 347, are designed to tie up carbon in more stable carbides before chromium can be depleted from the matrix.
This makes them far more resistant to sensitization than unstabilized grades in many welded or heat-exposed applications.
Select alloys matched to the environment
In aggressive chloride, Acid, or high-temperature service, it may be better to move away from susceptible families altogether and choose alloys with stronger grain-boundary stability, such as duplex stainless steels or nickel-based corrosion-resistant alloys.
Sa madaling salita, material selection should be based not only on base-metal strength, but also on how the alloy behaves after fabrication and during long-term exposure.
Heat Treatment Control: Manage the Microstructure, Not Just the Temperature
Heat treatment is one of the most powerful tools for preventing intergranular corrosion because it determines whether harmful grain-boundary precipitates form and remain in place.
Solusyon pagsusubo
For susceptible stainless steels, solusyon annealing is the standard corrective and preventive treatment.
The alloy is heated into the solution range so that precipitates dissolve back into the matrix, then cooled rapidly enough to prevent re-precipitation during the sensitive temperature range.
This restores a more uniform composition and helps recover corrosion resistance.
Rapid cooling after heating
The cooling rate is just as important as the peak temperature. Slow cooling through the sensitization range allows grain-boundary carbides or intermetallic phases to form.
Mabilis na paglamig, often by quenching when appropriate to the alloy and part geometry, helps preserve the solution-treated condition.
Post-weld init paggamot
For welded parts, post-weld heat treatment may be needed to reduce residual stress and restore a more favorable microstructure in the heat-affected zone.
The exact cycle depends on the alloy family, kapal ng seksyon, and service requirement.
The goal is not simply to “heat the part again,” but to eliminate the grain-boundary chemistry that makes the region vulnerable.
Welding Control: Keep the Heat-Affected Zone Out of Trouble
Welding is one of the most common causes of intergranular corrosion because it creates exactly the thermal conditions that promote grain-boundary precipitation and sensitization.
That is why welding practice must be tightly controlled.
Keep heat input as low as practical
High heat input enlarges the heat-affected zone and increases the time the material spends in the critical temperature range where harmful precipitation can occur.
Lower heat input helps reduce both the width and severity of the sensitized region.
Limit repeated thermal cycling
Multiple passes over the same region can intensify sensitization and enlarge the affected zone.
Welding procedures should minimize unnecessary reheating of previously welded areas.
Choose filler metals carefully
The filler metal should be compatible with the base alloy and should not introduce an unnecessary carbon or composition imbalance.
In susceptible stainless steels, low-carbon or stabilized filler systems are often preferred so the weld zone does not become the weak point.
Control cooling after welding
Rapid cooling helps the weld region move quickly through the danger zone where precipitates form.
The cooling method must be chosen carefully so it does not introduce distortion or cracking, but the basic principle remains the same: do not allow the heat-affected zone to linger in the sensitization range.
Environmental Control: Reduce the Driving Force for Attack
Even a susceptible microstructure may remain acceptable if the service environment is mild.
Sa kabilang banda, a moderate alloy can fail quickly in a severe environment.
That is why environmental control is a critical part of intergranular corrosion prevention.
Reduce exposure to aggressive media
Limit contact with acids, Mga klorido, or other corrosive species whenever possible.
In process systems, this may mean changing chemistry, lowering temperature, or reducing stagnation and concentration effects.
Control oxygen and moisture where relevant
In aqueous systems, dissolved oxygen and unfavorable electrochemical conditions can accelerate corrosion reactions.
Deoxygenation or chemistry control may help reduce the driving force for attack in susceptible systems.
Use coatings or linings when appropriate
Protective coatings, polymer linings, or internal barriers can isolate the alloy from the corrosive environment.
This is especially useful when the base alloy must be retained for mechanical reasons but the environment is too aggressive for bare metal.
Apply cathodic protection in suitable systems
For some structures, cathodic protection can reduce the electrochemical tendency toward corrosion.
This is not a universal solution, but in the right environment it can be an effective part of a larger corrosion-control program.
Paggamot sa ibabaw: Restore and Protect the Passive State
The surface condition of a component strongly influences its corrosion performance, especially after fabrication or welding.
Passivation
Passivation is used to clean the surface and promote a more stable passive film. It helps remove free iron and other contaminants that can interfere with corrosion resistance.
Email Address *
Pickling removes oxide scale, heat tint, and other surface contaminants, especially after welding or thermal exposure.
This matters because a damaged or contaminated surface can become the starting point for localized attack even when the internal microstructure is otherwise acceptable.
Electropolishing
Electropolishing smooths the surface and can improve passive-film uniformity.
By reducing roughness and surface irregularities, it can also reduce local sites where corrosion is more likely to initiate.
7. Test Methods and Applications
| Pamantayan / paraan ng | Material family | What it tells you | Typical use |
| ASTM A262 | Austenitic hindi kinakalawang na asero | Screens susceptibility to intergranular attack with oxalic acid etch, ferric sulfate-sulfuric acid, nitric acid, and copper/copper-sulfate methods. | Material qualification, sensitization screening, failure analysis. |
| ASTM A763 | Ferritic hindi kinakalawang na asero | Detects susceptibility to intergranular attack using practices W, X, Y, at Z. | Ferritic grade qualification and weld/heat-treatment assessment. |
ASTM G108 |
AISI Type 304 / 304L | Quantitatively measures degree of sensitization by electrochemical reactivation. | Research, comparative sensitization ranking, process verification. |
These standards are useful because intergranular corrosion is often invisible until damage is well advanced.
ASTM A262 is therefore a practical screen for austenitic stainless materials, ASTM A763 serves the ferritic family, and ASTM G108 gives a quantitative sensitization metric for 304 and 304L.
Used together, they allow the metallurgist to separate “apparently acceptable” from “actually resistant.”
8. Integration into an Integrity Management System
A robust integrity management system should treat intergranular corrosion as a life-cycle control problem, not just a material-test problem.
Sa pagsasanay, that means alloy qualification, welding procedure control, heat-treatment records, periodic inspection,
and failure-analysis feedback should all be tied together so that sensitization does not re-enter the system unnoticed.
This is an engineering inference from the way ASTM A262, ASTM A763, and ASTM G108 are used to screen materials and quantify sensitization before field failure occurs.
For critical equipment, the most effective approach is to connect material selection, fabrication history, and service environment into one control loop.
If a part is stainless, the question is not only whether it is stainless but whether it was welded, Init na ginagamot, and cleaned in a way that preserved chromium-rich passivity at the grain boundaries.
If it is aluminum or nickel alloy, the question is whether the precipitate structure or grain-boundary segregation has been pushed into a corrosive state.
That system-level view is what keeps IGC from becoming a hidden life-limiting mechanism.
9. Pangwakas na Salita
Intergranular corrosion is a grain-boundary corrosion mode driven by local chemistry, precipitation, segregasyon, and thermal history.
It is dangerous because it can remove strength and integrity while leaving the surface deceptively intact.
The mechanism is well understood in austenitic stainless steels, but it also appears in ferritic stainless steels, duplex hindi kinakalawang na asero, age-hardenable aluminum alloys, and nickel-based alloys when grain-boundary chemistry becomes unfavorable.
The practical defense is equally clear: choose the right alloy, control heat input and cooling history, validate with the correct ASTM test method, and treat the heat-affected zone as a critical quality feature.
Intergranular corrosion is not just a corrosion problem; it is a metallurgy, gawa gawa lang, and reliability problem.
Mga FAQ
What is the difference between intergranular corrosion and general corrosion?
General corrosion attacks the surface more or less uniformly,
while intergranular corrosion follows grain boundaries and can cause severe internal weakening with relatively little visible surface loss.
Why are stainless steels so often discussed in intergranular corrosion?
Because many stainless steels, especially austenitic grades, can become sensitized when chromium carbides form at grain boundaries and leave chromium-depleted zones behind.
ASTM A262 exists specifically to detect this susceptibility.
Can welding cause intergranular corrosion?
Oo nga. Welding can create a heat-affected zone that spends time in the sensitization range, promotes precipitates or segregation,
and leaves heat tint or other surface conditions that reduce corrosion resistance.
How do low-carbon stainless grades help?
Lower carbon reduces the driving force for chromium carbide precipitation,
and grades such as 304L, 316L, 317L, 321, at 347 are specifically used to resist sensitization during ordinary welding operations.